|
Thompson describes the critical experiments like this:Īs the cathode rays carry a charge of negative electricity, they are deflected by an electrostatic force as if they were negatively electrified, and are acted on by a magnetic force in just the way in which this force would act on a negatively electrified body moving along the path of these rays, I can see no escape from the conclusion that they are charges of negative electricity carried by particles of matter. Once he had this apparatus further experiments with electric and magnetic fields could reveal a great deal more about the particles. 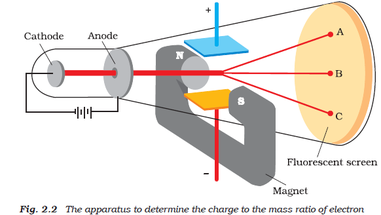
This enabled Thompson to refute the claim by Hertz that electric fields didn't cause deflection (maybe Hertz's glassblower wasn't as good). The critical experiments done by Thompson relied on having an excellent glassblower who was able to construct an apparatus where electrodes inside the discharge tube which could be charged with an electric field (while retaining a very high vacuum). Hertz, though, claimed electric fields would not deflect them (he was wrong). Later, scientists such as Crookes observed that the rays could be deflected by magnetic fields (though he thought they were just negatively charged gas particles). 
Initially the rays were seen to travel in straight lines. So the idea that some sort of ray was emitted by the cathode in the tube was developed. They were first observed as experiments in gas discharge tubes started to exploit better and better vacuums (the early experiments observed the varying forms of discharge in low pressure gases cathode rays only become significant when there is very little gas left in the tubes).Īt very high vacuums, people observed that the glass at the end of the tubes would glow even though the contents of the tubes did not (as there were too few gas particles left to cause a discharge). When the rays hit the paddle surface they heated it, and the heat caused the gas next to it to expand, pushing the paddle.Thompson was able to measure the ratio of their mass to their speedĬathode "rays" had been known for some time before Thomson. However, later it was concluded that the paddle wheel turned not due to the momentum of the particles (or electrons) hitting the paddle wheel but due to the radiometric effect (it is the repulsive force between two surfaces maintained at different temperatures). Crookes concluded at the time that this showed that cathode rays had momentum, so the rays were likely matter particles. The paddlewheel turned in a direction away from the cathode side of the tube, suggesting that the rays were coming from the cathode. So you should place it different tube to get the paddle wheel move by anode or cathode rays.Ĭrookes put a tiny vaned turbine or paddlewheel in the path of the cathode rays, and found that it rotated when the rays hit it. 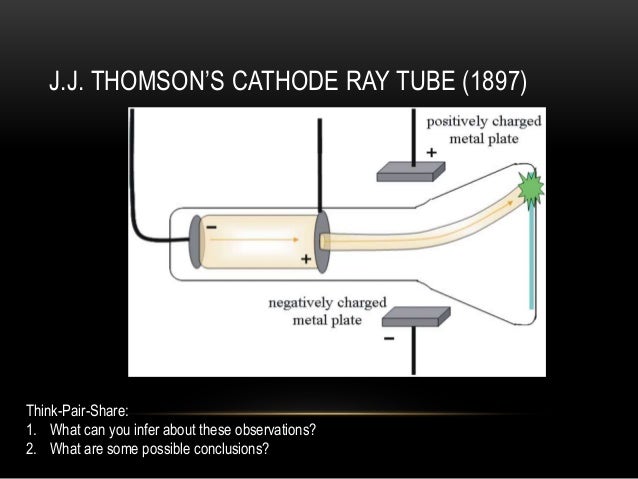
However, the tube must be set up quite differently from the usual cathode ray set-up in order to detect canal (or anode) rays. When high voltage is applied on electrodes the cathode emits electrons which collide with the residual gas knocking of electron from that gas molecule which travel towards the anode and the positively charged gaseous atom gaseous atom moves back to cathode which is anode rays. It is produced it the middle of the tube. Actually anode rays are not emitted from anode and reaching cathode.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
